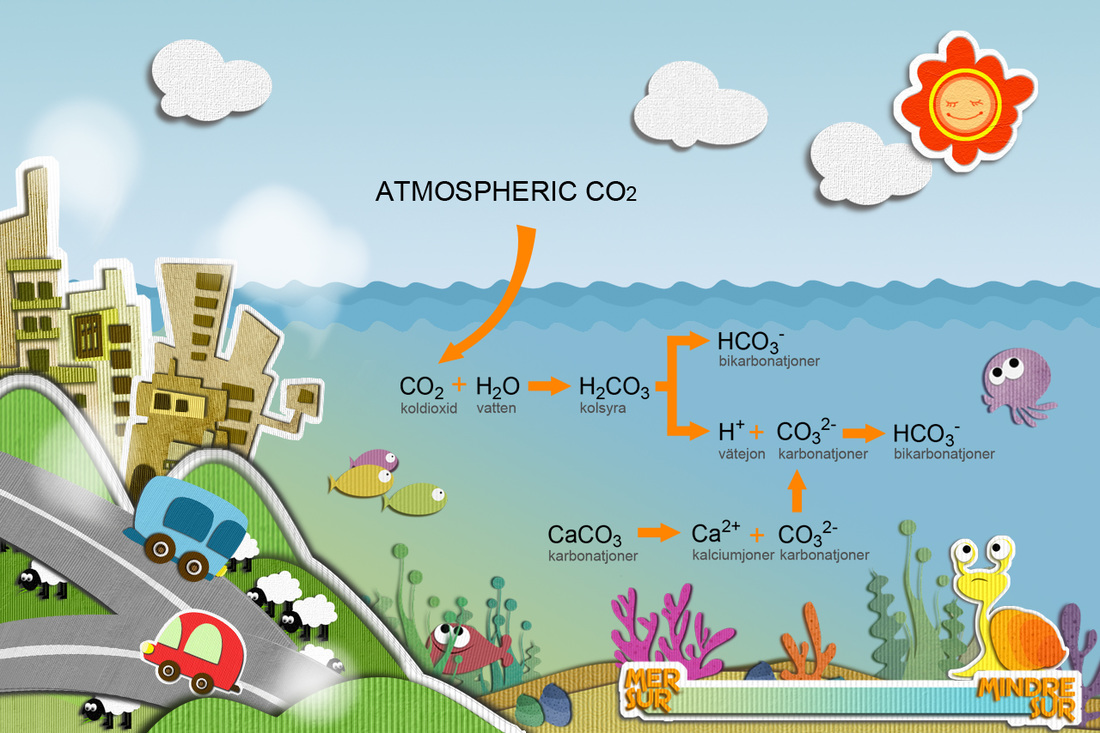
Oceans are a source of food, transportation climate regulation, and half of the Earth's oxygen to the human population.The oceans of the world have absorbed almost half of the CO2 emitted by humans from the burning of fossil fuels, causing a decrease in the PH of the ocean,forming carbonic acid, This is called ocean acidification, which affects all living organisms that rely on shells. Carbonic acid causes the shells to dissolve.
METHODS AND MATERIALS:Protocol: There were 4 different protocols to follow. Three experimental protocols and one control protocol. The experimental protocols were room temperature tap water, cold salt water, and hot salt water. The control protocol was room temperature salt water(D).
Tap and Control
Universal indicator was dropped into protocols in order to determine pH levels by recording the initial color. A straw was used to blow bubbles at a constant rate and then recorded the color of the solution every 30 seconds for 2 minutes.
Cold Salt Water
Beaker was placed on ice for approximate 3 minutes before the process of blowing and recording every 30 second for 2 minutes.
Hot Salt Water
Protocol was placed on a hot plate for 3 minutes and then the same process as tap and control protocols was done.
RESULTS:CONTROL
0 Seconds 30 Seconds 60 Seconds 90 Seconds 120 Seconds
7.8 pH, Dark green 7.3 pH, Green 6.5 pH, Light green 6.6 pH, Light green 6.6 pH, Light green
COLD
0 Seconds 30 Seconds 60 Seconds 90 Seconds 120 Seconds
7.9 pH, Dark green 7.6 pH, Dark green 7.0 pH, Green 6.5 pH, Light green 6.4 pH, Light green
Hot
0 Seconds 30 Seconds 60 Seconds 90 Seconds 120 Seconds
8.1 pH, Dark green 7.2 pH, Green 7.0 pH, Green 7.0 pH, Green 6.8 pH, Light green
Tap
0 Seconds 30 Seconds 60 Seconds 90 Seconds 120 Seconds
7.8 pH, Dark green 6.8 pH, Light green 6.5 pH, Light green 6.4 pH, Light green 6.2 pH, Yellow
Tap and Control
Universal indicator was dropped into protocols in order to determine pH levels by recording the initial color. A straw was used to blow bubbles at a constant rate and then recorded the color of the solution every 30 seconds for 2 minutes.
Cold Salt Water
Beaker was placed on ice for approximate 3 minutes before the process of blowing and recording every 30 second for 2 minutes.
Hot Salt Water
Protocol was placed on a hot plate for 3 minutes and then the same process as tap and control protocols was done.
RESULTS:CONTROL
0 Seconds 30 Seconds 60 Seconds 90 Seconds 120 Seconds
7.8 pH, Dark green 7.3 pH, Green 6.5 pH, Light green 6.6 pH, Light green 6.6 pH, Light green
COLD
0 Seconds 30 Seconds 60 Seconds 90 Seconds 120 Seconds
7.9 pH, Dark green 7.6 pH, Dark green 7.0 pH, Green 6.5 pH, Light green 6.4 pH, Light green
Hot
0 Seconds 30 Seconds 60 Seconds 90 Seconds 120 Seconds
8.1 pH, Dark green 7.2 pH, Green 7.0 pH, Green 7.0 pH, Green 6.8 pH, Light green
Tap
0 Seconds 30 Seconds 60 Seconds 90 Seconds 120 Seconds
7.8 pH, Dark green 6.8 pH, Light green 6.5 pH, Light green 6.4 pH, Light green 6.2 pH, Yellow
Conclusion:
The more bubbles that were blown the more acidic the water became. Based on the results, the warmer the water the more acidic it becomes.
The more bubbles that were blown the more acidic the water became. Based on the results, the warmer the water the more acidic it becomes.
The Effect of Acidifiaction on Shells Mass
Methods and Materials
Two untreated shells labeled, "E" for experimental and "C" for control. The initial characteristics of both the shells were recorded and massed. The vinegar was used to resemble acid in the water and hoe the shells are affected by the "acid". Shell "C" was placed in salt water and shell "E" to the beaker of vinegar for 30 minutes. The shell in vinegar started reacting to the vinegar, forming bubbles and was noted that it started peeling. Observations of shell characteristics for the pre-treated ("Low Exposure" and "High Exposure") shells were also recorded at the same time as "C" and "E". At the end all four shells were re-massed, After textbooks were used to observe how strong shells "C" and "E" were.
Two untreated shells labeled, "E" for experimental and "C" for control. The initial characteristics of both the shells were recorded and massed. The vinegar was used to resemble acid in the water and hoe the shells are affected by the "acid". Shell "C" was placed in salt water and shell "E" to the beaker of vinegar for 30 minutes. The shell in vinegar started reacting to the vinegar, forming bubbles and was noted that it started peeling. Observations of shell characteristics for the pre-treated ("Low Exposure" and "High Exposure") shells were also recorded at the same time as "C" and "E". At the end all four shells were re-massed, After textbooks were used to observe how strong shells "C" and "E" were.
Results
Shells Initial Mass Final Mass Difference Observations
____________________________________________________________________________________________
(L) exposure 3.633 grams 3.665 grams -0.032 grams 3 txt books to break
____________________________________________________________________________________________
(H) exposure 4.799 grams 4.856 grams -0.057 grams 3.5 txt books to break
____________________________________________________________________________________________
E 2.011 grams 1.892 grams 0.119 grams 3 txt books to break
____________________________________________________________________________________________
C 2.042 grams 2.450 grams -0.408 grams 3 txt books to break
____________________________________________________________________________________________
(L) exposure 3.633 grams 3.665 grams -0.032 grams 3 txt books to break
____________________________________________________________________________________________
(H) exposure 4.799 grams 4.856 grams -0.057 grams 3.5 txt books to break
____________________________________________________________________________________________
E 2.011 grams 1.892 grams 0.119 grams 3 txt books to break
____________________________________________________________________________________________
C 2.042 grams 2.450 grams -0.408 grams 3 txt books to break
Conclusion
According to the results it is evident that vinegar makes sea shells weaker, causing them to dissolve. The weaker the shells the less organisms can live because they are dependent of the shells to survive in the ocean.